  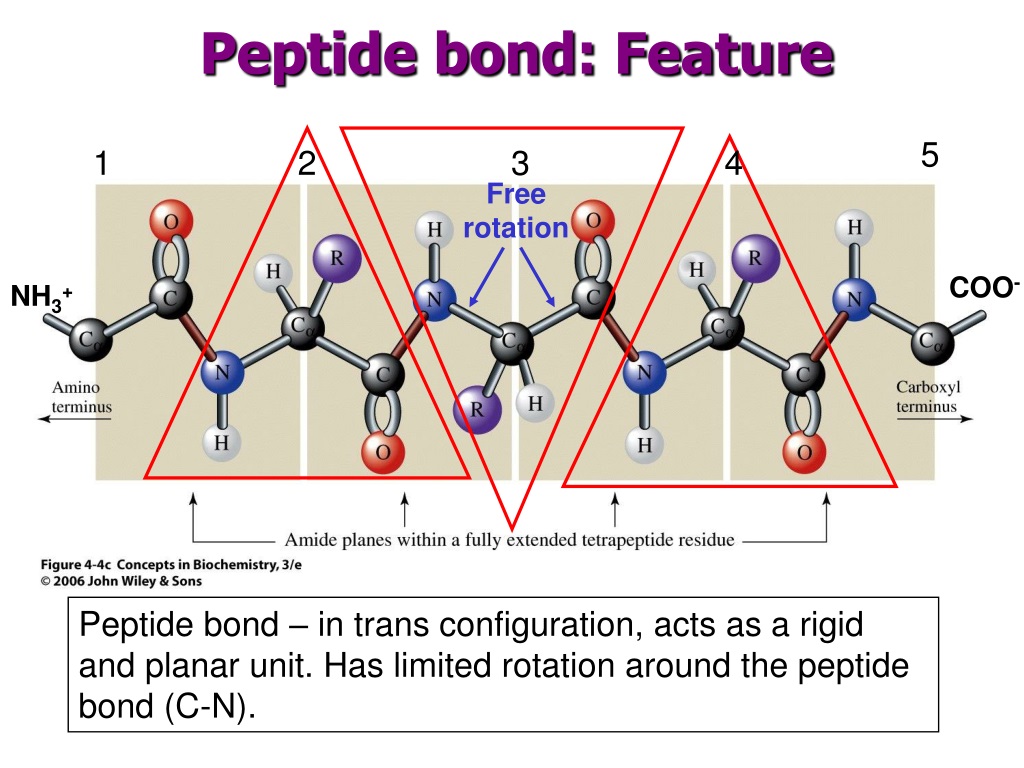
The BAL anchoring is established by anchoring an aromatic aldehyde, typically a trisalkoxybenzaldehyde, to the solid support, followed by attachment of the first amino acid residue by reductive amination. The BAL strategy was originally developed using a trisalkoxybenzyl linker, but since then range linkers (handles) with different properties have also been developed. This is thus a very general strategy for the introduction of C-terminal modifications. Cis and trans isomers are at far left and far right, respectively, separated by the transition states.In the backbone amide linker (BAL) strategy, the peptide is anchored not at the C-terminus but through a backbone amide, which leaves the C-terminal available for various modifications. However, X-Pro peptide groups tend to have a roughly 30:1 ratio, presumably because the symmetry between the C α and C δ atoms of proline makes the cis and trans isomers nearly equal in energy, see the figure. The trans form is preferred overwhelmingly in most peptide bonds (roughly 1000:1 ratio in trans:cis populations). In the unfolded state of proteins, the peptide groups are free to isomerize and adopt both isomers however, in the folded state, only a single isomer is adopted at each position (with rare exceptions). The partial double bond renders the amide group planar, occurring in either the cis or trans isomers. Significant delocalisation of the lone pair of electrons on the nitrogen atom gives the group a partial double-bond character. The wavelength of absorption for a peptide bond is 190–230 nm, which makes it particularly susceptible to UV radiation.Ĭis/trans isomers of the peptide group This non-enzymatic process is thus not accelerated by transition state stabilization, but rather by ground-state destabilization. In living organisms, the process is normally catalyzed by enzymes known as peptidases or proteases, although there are reports of peptide bond hydrolysis caused by conformational strain as the peptide/protein folds into the native structure. This process is extremely slow, with the half life at 25 ☌ of between 350 and 600 years per bond. The hydrolysis of peptide bonds in water releases 8–16 kJ/ mol (2–4 kcal/ mol) of Gibbs energy. Degradation Ī peptide bond can be broken by hydrolysis (the addition of water). For example, the tripeptide glutathione is synthesized in two steps from free amino acids, by two enzymes: glutamate–cysteine ligase (forms an isopeptide bond, which is not a peptide bond) and glutathione synthetase (forms a peptide bond). Some peptides, like alpha-amanitin, are called ribosomal peptides as they are made by ribosomes, but many are nonribosomal peptides as they are synthesized by specialized enzymes rather than ribosomes. Organisms use enzymes to produce nonribosomal peptides, and ribosomes to produce proteins via reactions that differ in details from dehydration synthesis. Peptides and proteins are chains of amino acids held together by peptide bonds (and sometimes by a few isopeptide bonds). The formation of the peptide bond consumes energy, which, in organisms, is derived from ATP. The dehydration condensation of two amino acids to form a peptide bond (red) with expulsion of water (blue) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
